|
11/18/2023 0 Comments Johnson and johnson vaccine production
–CNBC's Nate Rattner contributed to this report. The only Johnson & Johnson facility currently making usable batches of the company’s single-shot coronavirus vaccine has temporarily halted its production, a new report said Tuesday. J&J reported last month that new research found its vaccine was effective against the highly contagious delta, even eight months after inoculation. health officials have maintained that the one-dose vaccine is safe and highly effective, especially against severe disease, hospitalizations and death. "I think the public hears that the vaccine is taken off the market for a period of time and it is just hard to get past that scarlet letter," said Offit, also director of the Vaccine Education Center at Children's Hospital of Philadelphia. 
officials determined that the benefits of the inoculations outweighed their risks. The recommended pause was lifted 10 days later after U.S. Paul Offit, who has served on advisory panels for both the CDC and the Food and Drug Administration, said J&J's vaccine really "suffered" after federal health officials in April asked states to temporarily halt using the shot "out of an abundance of caution" while it investigated six women who developed a rare, but serious, blood-clotting disorder. Personal Loans for 670 Credit Score or Lowerĭr. Personal Loans for 580 Credit Score or Lower up to 15 million doses of Johnson & Johnson’s vaccine and forcing regulators to delay authorization of the plant’s production lines. DNA is not as fragile as RNA, and the adenovirus’s tough protein coat helps protect the genetic. Johnson & Johnson’s vaccine is delayed by a U.S. 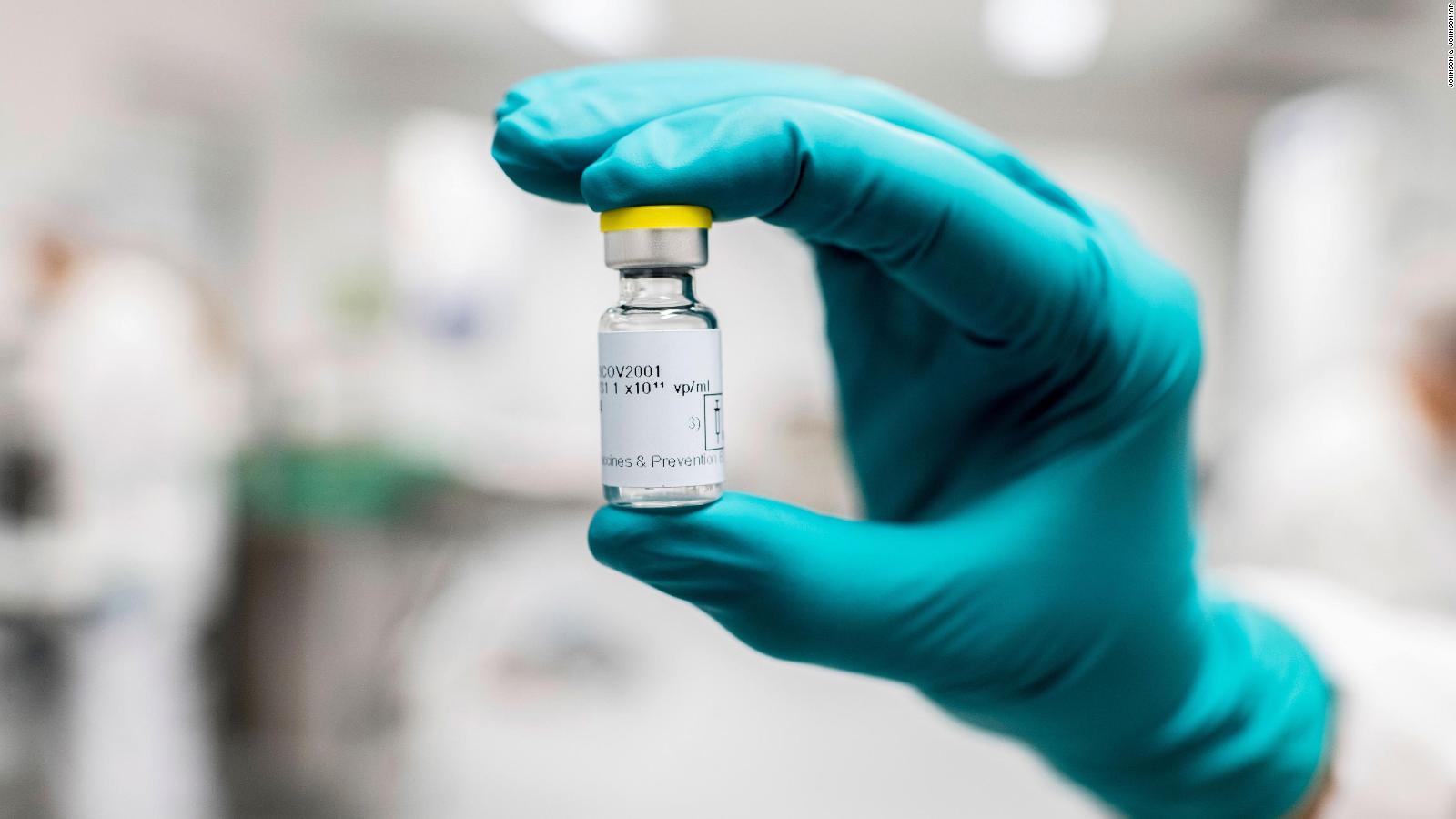
Although it will take studies in people to be certain the vaccines will work against variants, "Anything that generates a T-cell immune response to the SARS-CoV-2, I would say has promise as being potentially protective," Maus says.Best Debt Consolidation Loans for Bad Credit Adenovirus-based vaccines for Covid-19 are more rugged than mRNA vaccines from Pfizer and Moderna. The one-dose J&J vaccines were exported from South Africa, where they had been assembled, despite the pressing need for vaccines across Africa. So has Marcela Maus at Massachusetts General Hospital. Sette's lab has had similar results with the Pfizer-BioNTech and Moderna COVID-19 vaccines. "Dan Barouch's data really show very nicely that there is no appreciable decrease in reactivity." "The data is very solid," says Alessandro Sette, an immunologist at the La Jolla Institute for Immunology. That may help explain why the Johnson & Johnson vaccine prevented serious disease when tested in volunteers South Africa, where worrisome variants are circulating. It's not just the Beta variant, but also the Alpha and Gamma variants. "The T-cell responses actually are not reduced - at all - to the variants," Barouch says. They don't prevent infection they help keep an infection from spreading. Recombinant vaccines use a small piece of genetic. The vaccine is also sometimes referred to as. It is a viral vector vaccine that uses a weakened live pathogen (adenovirus) as the delivery method (vector) for transporting a recombinant vaccine for COVID-19. Johnson & Johnson vaccine was contaminated by ingredients from another company’s vaccine at a manufacturing plant in Baltimore, federal officials confirmed Wednesday, ruining a batch of raw. "Those are the types of T cells that can basically seek out and destroy cells that are infected and help clear infection directly." Johnson & Johnson's vaccine is a single-dose adenovirus-based vaccine. "Those are the killer T cells," Barouch says. (Johnson & Johnson) uses a first-generation Ad26 vector (E1/E3 deleted) to deliver the pre-fusion stabilized SARS-CoV-2. Shots - Health News Got Questions About Johnson & Johnson's COVID-19 Vaccine? We Have Answers The Ad26.COV2-S vaccine developed by Janssen Vaccines & Prevention B.V. Under the new nomenclature proposed by the World Health Organization, B.1.351 is now called Beta. Under its agreement with the US government, Johnson & Johnson was targeted to produce twelve million doses by the end of February 2021, more than sixty million. "What we showed is that the neutralizing antibodies are reduced about fivefold to the B.1.351 variant," says Dan Barouch, director of the Center for Virology and Vaccine Research at Beth Israel Deaconess Medical Center in Boston. Scientists took blood from volunteers who had received the Johnson & Johnson COVID-19 vaccine and looked at the levels of neutralizing antibodies, the kind that prevent a virus from entering cells. The emergence of new and more infectious variants of the coronavirus has raised a troubling question: Will the current crop of COVID-19 vaccine prevent these variants from causing disease?Ī study out Wednesday in the journal Nature suggests the answer is yes. A woman receives the Johnson & Johnson COVID-19 at a drive-in vaccination event last week in Meerbusch, Germany.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
